Loading...
Loading...
Deviation Management Software helps pharmaceutical and life sciences companies record, track, and close deviations effectively. It ensures compliance with industry regulations and provides a structured way to handle both planned and unplanned deviations. With secure electronic records, approvals, and reminders, it makes the deviation process faster, transparent, and compliant. It integrates with CAPA and Investigation module.
Our Deviation Management Software is designed with pharmaceutical quality standards in mind. It supports full deviation lifecycle management — from initiation to root cause analysis and closure. The system is web-based, user-friendly, and compliant with 21 CFR Part 11 (USFDA) requirements. Site QA teams can easily track progress, monitor timelines, and ensure corrective actions are properly implemented.
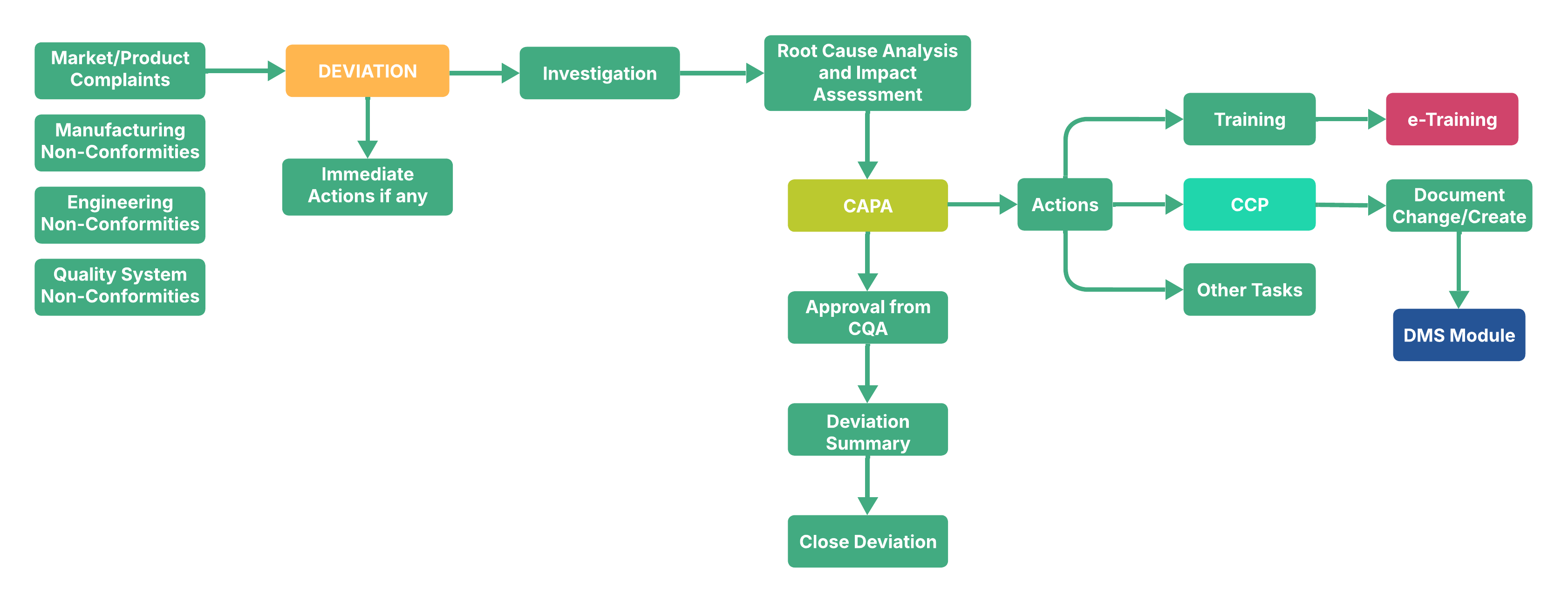
By using Deviation Management Software, pharma and life sciences organizations can reduce manual effort, avoid missed deadlines, and ensure regulatory compliance. It simplifies deviation handling while maintaining accuracy, accountability, and transparency across departments.